US FDA 21 CFR Part 820 (QSR)
Labquality has great expertise with quality systems, including the USA Food and Drug Administration Quality System Regulation, QSR, 21 CFR Title 820.
What are the QSR requirements?
Any company that want to sell their medical devices in the United States has to be compliant to FDA quality system regulation 21 CFR Part 820 (QSR). The QSR set the requirements for quality management system and are similar to ISO 13485 requirements. However, there are certain differences. The FDA QSR is a US law that needs to be followed, whereas ISO 13485 status is not similar in the EU.
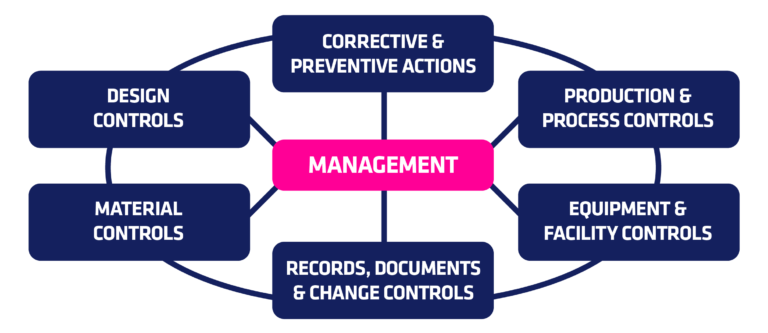
Quality System Regulation 21 CFR 820
According to the regulation, manufacturers shall
- Establish that the quality system is consistent with the complexity of the device, manufacturing processes and size, and the complexity of the manufacturing facility
- Plan to define and implement effective procedures
- Implement what has been documented and is going to be done
- Check the system and make necessary changes (CAPA)
- Act upon changes and ensure they are implemented
.jpg)
Labquality
How can we help?
Labquality can assist you onPlanning the QMS together with the manufacturer according to the QSR regulation
Helping the manufacturer identify all processes needed for their QSR
Preparing the Quality Manual, QMS process descriptions, templates, databases, and other documentation needed together with the manufacturer
Helping the manufacturer choose and implement electronic QMS when needed
Helping the manufacturer maintain and continually improve their QMS
Support the manufacturer with daily QMS activities including nonconformances, feedback, complaints, internal and supplier audits, vigilance, and many others
Offering customized and open training for medical device QMS

Contact us for more information
Leave a contact request
Related Services

QMS Improvement and Gap Analysis
Labquality can help medical device manufacturers continually improve their quality management system (QMS).

Quality Management System ISO 13485
Labquality provides medical device manufacturers help with setting up quality management systems (QMS).
Latest news
Meet us in Tomar, Portugal
Labquality will attend SPML's (Sociedade Portuguesa de Medicina Laboratorial) 16th scientific meeting in Tomar, Portugal, from 12 to 13 April 2024.Meet us at ECCMID in Barcelona
Visit our booth E5 in the ECCMID exhibition and hear about EQA services for microbiology, as well as CRO services for pharmaceuticals and MD/IVD manufacturers.Why Modeling Data Flows Is Needed in Clinical Investigations
data management manager explains how data flow modeling will help keep clinical investigation stakeholders informedEQA Coordinator Anna-Riitta Vanhanen Retires After 34 Years of Service
EQA Coordinator Anna-Riitta Vanhanen is retiring in the autumn of 2024 after working at Labquality since 1990. Biochemist Hanna Heikkilä will take over Anna-Riitta's EQA rounds.Read now the new customer reference
Peili Vision has chosen Labquality as the partner for a clinical study to assess the performance and safety of their browser-based, game-like medical software.Subscribe to our newsletter
Subscribe to hear the latest news in the industry and keep track of what's happening behind the scenes.




