Risk Management ISO 14971
Labquality provides medical device manufacturers help in setting up and implementing a risk management system and preparing risk management documentation for their medical products.
Risk management with medical devices
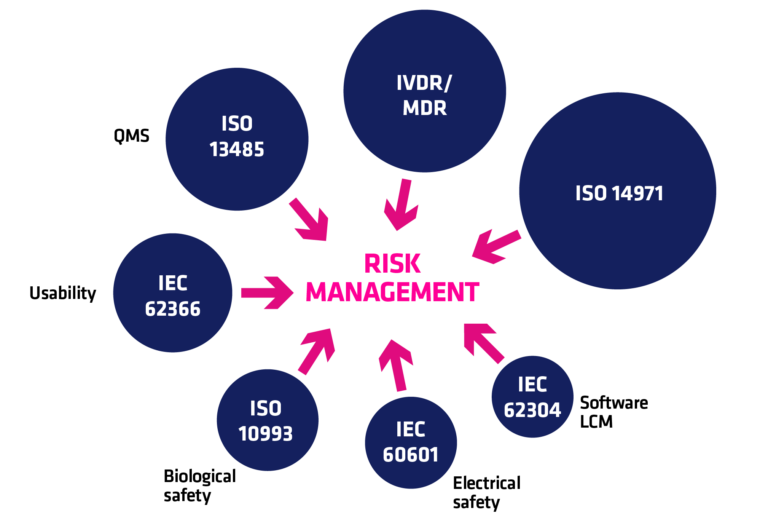
Risk management is the core element of medical device regulations. You must have heard about the "risk-based approach" when discussing medical devices or their quality management systems. Medical device risk management is most conveniently performed following ISO 14971, which is harmonized with Regulations 2017/745 (MDR) and 2017/746 (IVDR) and is US FDA recognised consensus standard.
Performing risk analysis is a process which produces a risk management file for each medical device. It consists of a risk management plan, risk analyses and risk management report. It should cover a wide array of different risk categories such as cybersecurity risks and usability risks.
Our experts at Labquality have long-term experience in risk management and its different flavours and can support you in creating efficient and compliant risk management operations in your company.
Labquality
How can we help?
Writing the risk management process description in accordance with applicable standards and regulations
Helping the manufacturer to implement and maintain the risk management process
Preparing risk management plans and reports
Helping the manufacturer with risk analysis covering all needed aspects (e.g., mfg processes, quality processes, design, usability, software, cybersec.)
Helping the manufacturer with benefit-risk determination
Helping the manufacturer in answering the risk management related questions raised by notified bodies and authorities
Offering customized and open training for medical device risk management

Contact us for more information
Leave a contact request
Related Services

Electrical Safety IEC 60601
IEC 60601-1 is the basic safety standard for medical electrical devices and systems.

Biological Safety ISO 10993
We provide medical device manufacturers help with setting up a biological safety evaluation process and preparing biological evaluation documentation.

IVD Performance Evaluation
Labquality helps manufacturers comply with the clinical evidence requirements of the IVD Regulation.

Qualification and classification of medical devices
Our team’s versatile experience and the available tools help in bringing clarity to the challenging cases.

Regulatory plan
Labquality can help medical device manufacturers in creating a regulatory plan.

IVDR compliance for in-house developed tests
Laboratories developing IVDs in-house must maintain Technical Documentation and Quality Management Systems to comply with the IVDR and the national law.

Medical software
Labquality also provides comprehensive support for medical software lifecycle.
Latest news
Meet us at RAPS in Berlin on May 6-8!
Visit our booth TB1 in the RAPS exhibition and hear about regulatory affairs services for and MD/IVD manufacturers.Meet us in Tomar, Portugal
Labquality will attend SPML's (Sociedade Portuguesa de Medicina Laboratorial) 16th scientific meeting in Tomar, Portugal, from 12 to 13 April 2024.Meet us at ECCMID in Barcelona
Visit our booth E5 in the ECCMID exhibition and hear about EQA services for microbiology, as well as CRO services for pharmaceuticals and MD/IVD manufacturers.Why Modeling Data Flows Is Needed in Clinical Investigations
data management manager explains how data flow modeling will help keep clinical investigation stakeholders informedEQA Coordinator Anna-Riitta Vanhanen Retires After 34 Years of Service
EQA Coordinator Anna-Riitta Vanhanen is retiring in the autumn of 2024 after working at Labquality since 1990. Biochemist Hanna Heikkilä will take over Anna-Riitta's EQA rounds.Subscribe to our newsletter
Subscribe to hear the latest news in the industry and keep track of what's happening behind the scenes.




